Atomic structure
Understand that atoms are the basic building blocks of all matter
Task 1
Take pictures of 3 examples of matter around the school in 5 minutes and label them, are they made of atoms? Insert your pictures below.
Recall the symbols of common elements
Periodic Table http://www.ptable.com/
Element song! http://www.privatehand.com/flash/elements.html
Task 2
Learn the names and symbols of the first 20 elements on the periodic table and some of the other common elements – Iron, Copper, Zinc, Silver, Bromine, Iodine, Gold, Mercury, Lead, Platinum, Uranium.
Construct a table with the 31 elements mentioned above with their name and symbol.
Task 3
TRY THIS FIRST 20 ELEMENTS TEST
http://chemistry.about.com/od/testsquizzes/l/blsymbolquiz.htm
Task 4
Try this quizlet link to test and extend your understanding
http://quizlet.com/11257964/element-names-flash-cards/
Definition of Element: X
For example: Oxygen, as well as all those on the periodic table.
Task 5
Develop a poster of an element. Choose an element that no one else in your class is researching, find out its uses, picture, discovery, and any other interesting facts ( physical properties) and make an electronic poster/brochure. Then copy and paste it in the area below (apps like pic collage will do). My chosen element is X
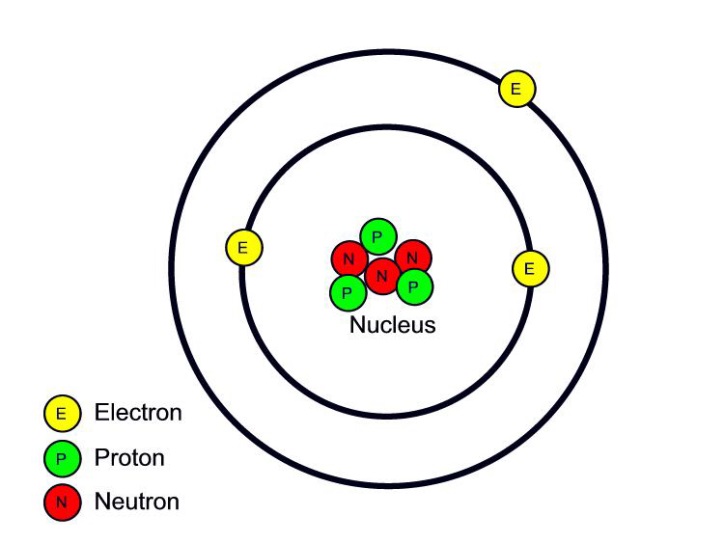
The structure of an atom
Although the word 'atom' comes from the Greek for indivisible, we now know that atoms are not the smallest particles of matter. Atoms are made from smaller subatomic particles.
Task 6
Look at the picture below and complete this table
Sub-atomic particles Location Charge Relative Mass
Proton X X X
Neutron X X X
Electron X X 1/1820
ELECTRONS ARE 1820 TIMES SMALLER than the protons and neutrons so we ignore the mass.
Task 1
Take pictures of 3 examples of matter around the school in 5 minutes and label them, are they made of atoms? Insert your pictures below.
Recall the symbols of common elements
Periodic Table http://www.ptable.com/
Element song! http://www.privatehand.com/flash/elements.html
Task 2
Learn the names and symbols of the first 20 elements on the periodic table and some of the other common elements – Iron, Copper, Zinc, Silver, Bromine, Iodine, Gold, Mercury, Lead, Platinum, Uranium.
Construct a table with the 31 elements mentioned above with their name and symbol.
Task 3
TRY THIS FIRST 20 ELEMENTS TEST
http://chemistry.about.com/od/testsquizzes/l/blsymbolquiz.htm
Task 4
Try this quizlet link to test and extend your understanding
http://quizlet.com/11257964/element-names-flash-cards/
Definition of Element: X
For example: Oxygen, as well as all those on the periodic table.
Task 5
Develop a poster of an element. Choose an element that no one else in your class is researching, find out its uses, picture, discovery, and any other interesting facts ( physical properties) and make an electronic poster/brochure. Then copy and paste it in the area below (apps like pic collage will do). My chosen element is X
The structure of an atom
Although the word 'atom' comes from the Greek for indivisible, we now know that atoms are not the smallest particles of matter. Atoms are made from smaller subatomic particles.
Task 6
Look at the picture below and complete this table
Sub-atomic particles Location Charge Relative Mass
Proton X X X
Neutron X X X
Electron X X 1/1820
ELECTRONS ARE 1820 TIMES SMALLER than the protons and neutrons so we ignore the mass.
Predict the number of protons, neutrons and electrons from the atomic number and mass number
Task 7
What do these two terms mean?
Atomic Number: X
Mass Number: X
Can you work out the number of Proton, Electrons and Neutron? ( PEN number)
Task 7
What do these two terms mean?
Atomic Number: X
Mass Number: X
Can you work out the number of Proton, Electrons and Neutron? ( PEN number)
Task 8
How many P,E,N does an atom of gold have?
P X
E X
N X
How do we work out the number of protons, neutrons and electrons? X (Explain clearly)
The type of element is determined by the number of X
Task 9
Complete the missing spaces:
Element name Atomic number Mass number Proton number Neutron number
X 2 4 X 2
X 7 14 7 X
neon 10 20 X X
X X X 3 4
X 20 X X 20
X X X 12 12
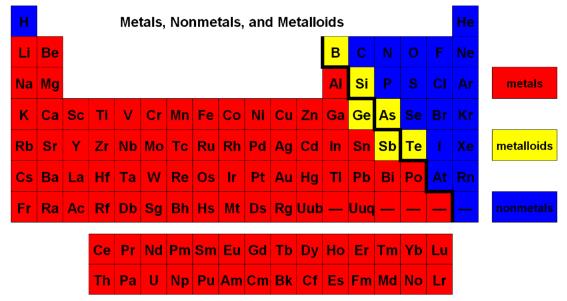
Distinguish between metals and non-metals using their position on the PT and their properties
The columns on the periodic table are called X
Group 1 all have 1 outer shell electron and are not stable, they want to lose 1 electron to have a full shell
The rows on the periodic table are called X
Elements on the same period have the number of electron shells
Properties of metal and non-metals
Look at the website
http://www.s-cool.co.uk/gcse/chemistry/metals-the-reactivity-series/revise-it/properties-of-metals-and-non-metals
How many P,E,N does an atom of gold have?
P X
E X
N X
How do we work out the number of protons, neutrons and electrons? X (Explain clearly)
The type of element is determined by the number of X
Task 9
Complete the missing spaces:
Element name Atomic number Mass number Proton number Neutron number
X 2 4 X 2
X 7 14 7 X
neon 10 20 X X
X X X 3 4
X 20 X X 20
X X X 12 12
Distinguish between metals and non-metals using their position on the PT and their properties
The columns on the periodic table are called X
Group 1 all have 1 outer shell electron and are not stable, they want to lose 1 electron to have a full shell
The rows on the periodic table are called X
Elements on the same period have the number of electron shells
Properties of metal and non-metals
Look at the website
http://www.s-cool.co.uk/gcse/chemistry/metals-the-reactivity-series/revise-it/properties-of-metals-and-non-metals
Where are the metals found on the periodic table? X
Where are the non-metals found on the periodic table? X
Where are the metalloids found on the periodic table? X
Task 10
Give 3 example of metals and non-metals each, then provide the uses of each element you have given.
Metal 1
Name X
Picture X
Uses X
Metal 2
Name X
Picture X
Uses X
Metal 3
Name X
Picture X
Uses X
Non-Metal 1
Name X
Picture X
Uses X
Non-Metal 2
Name X
Picture X
Uses X
Non-Metal 3
Name X
Picture X
Uses X
Task 11
Why do we use Iron for making cooking pans? X
Why do carbon and hydrogen compounds were used to make plastic handles for cooking pans? X
Why do we use copper for electrical wires? X
Where are the non-metals found on the periodic table? X
Where are the metalloids found on the periodic table? X
Task 10
Give 3 example of metals and non-metals each, then provide the uses of each element you have given.
Metal 1
Name X
Picture X
Uses X
Metal 2
Name X
Picture X
Uses X
Metal 3
Name X
Picture X
Uses X
Non-Metal 1
Name X
Picture X
Uses X
Non-Metal 2
Name X
Picture X
Uses X
Non-Metal 3
Name X
Picture X
Uses X
Task 11
Why do we use Iron for making cooking pans? X
Why do carbon and hydrogen compounds were used to make plastic handles for cooking pans? X
Why do we use copper for electrical wires? X