Chemicals and Chemical Reactions
Distinguish between atoms and molecules
Definition of Atom: X
Definition of Molecule: X
How is an atom different to a molecule? X
Task 12
Give 2 examples on molecules: X
Explain why they are molecules. X
Give 2 examples of things made of just atoms: X
Classify substances as elements, compounds and mixtures
An element cannot be broken down into simpler substances. Water can be broken down in to oxygen and hydrogen gases. So water is not an element.
Task 13
Read SW pg 168-169, Homework pg 169 activity
Most of the elements are metals. E.g. X
The rest are non-metals. E.g. X
Metalloids are between metal and non-metal. E.g X
Definition of a Compound: X
Definition of a Mixture: X
Definition of Atom: X
Definition of Molecule: X
How is an atom different to a molecule? X
Task 12
Give 2 examples on molecules: X
Explain why they are molecules. X
Give 2 examples of things made of just atoms: X
Classify substances as elements, compounds and mixtures
An element cannot be broken down into simpler substances. Water can be broken down in to oxygen and hydrogen gases. So water is not an element.
Task 13
Read SW pg 168-169, Homework pg 169 activity
Most of the elements are metals. E.g. X
The rest are non-metals. E.g. X
Metalloids are between metal and non-metal. E.g X
Definition of a Compound: X
Definition of a Mixture: X
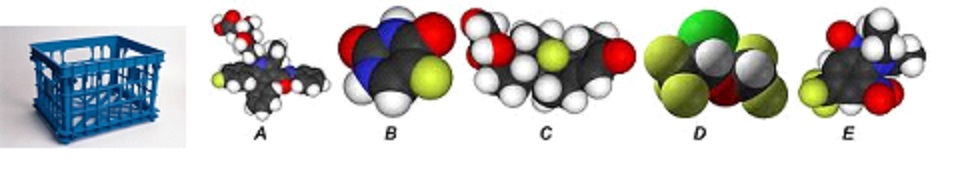
Is plastic an element, mixture or compound? X
How about A-E in the above picture? X
Task 14
Take pictures of 3 objects to show an example of an element, a mixture and a compound and list what they are made of.
An example on water is done for you
Object name: Water
Compound or element? Compound
What it's made of: 2 H atoms and 1 O atom
Picture:
Object name:
Compound or element?
What it's made of:
Picture:
Object name:
Compound or element?
What it's made of:
Picture:
Object name:
Compound or element?
What it's made of:
Picture:
Molecules are made of non-metals chemically bonded e.g. oxygen molecule, water molecule.
Which of these molecules is a compound? X
Which molecule is an element? X
Task 15
SW pg 171-172 exercises for homework
Distinguish between elements and compounds
Distinguish between mixtures and compounds
You may find these websites helpful to preview to complete the task below
http://www.bbc.co.uk/bitesize/ks3/science/chemical_material_behaviour/compounds_mixtures/revision/3/
http://www.bbc.co.uk/bitesize/ks3/science/chemical_material_behaviour/compounds_mixtures/revision/1/
Task 16
Try this Quiz online to ensure your understanding
http://quizlet.com/11278379/jc-science-chemistry-element-compound-mixture-check-list-chap-19-flash-cards/
Task 17
Go to this website and complete the worksheet
http://chemsite.lsrhs.net/Intro/handouts/Elements_compounds_mixtures.pdf
Interpret chemical formulae:
A chemical formulae tells you what elements are in the chemical, also the ratio of each element in the chemical.
For example the formula for carbon monoxide is CO. It tells you that each molecule of carbon monoxide consists of one carbon atom joined to one oxygen atom.
Take care when writing your symbols and formulae. Be careful about when to use capital letters. For example CO means a molecule of carbon
monoxide but Co is the symbol for cobalt.
Formula and formulae
The word 'formulae' ("form-u-lee") is the plural of 'formula'. If we have more than one formula, we don't say formulas, we say formulae.
Numbers in formulae
If the molecule contains more than one atom of an element we use numbers to show this. The numbers are written to the right and slightly below
the element symbol. For example, the formula for carbon dioxide is CO2 . It tells you that each molecule has one carbon atom and two oxygen atoms.
Take care when writing these formulae. The small number is written at the bottom right corner.
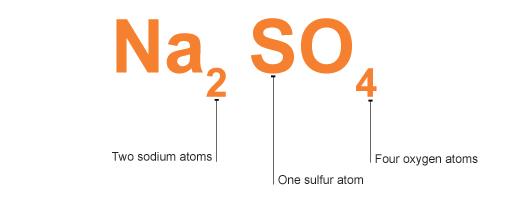
Some formulae are more complicated. For example, the formula for sodium sulphate is Na2SO4 .
It tells you that sodium sulphate contains two sodium atoms (Na2 ), one sulphur atom (S) and four oxygen atoms (O4 ).
Task 18
Try these exercises:
How about A-E in the above picture? X
Task 14
Take pictures of 3 objects to show an example of an element, a mixture and a compound and list what they are made of.
An example on water is done for you
Object name: Water
Compound or element? Compound
What it's made of: 2 H atoms and 1 O atom
Picture:
Object name:
Compound or element?
What it's made of:
Picture:
Object name:
Compound or element?
What it's made of:
Picture:
Object name:
Compound or element?
What it's made of:
Picture:
Molecules are made of non-metals chemically bonded e.g. oxygen molecule, water molecule.
Which of these molecules is a compound? X
Which molecule is an element? X
Task 15
SW pg 171-172 exercises for homework
Distinguish between elements and compounds
Distinguish between mixtures and compounds
You may find these websites helpful to preview to complete the task below
http://www.bbc.co.uk/bitesize/ks3/science/chemical_material_behaviour/compounds_mixtures/revision/3/
http://www.bbc.co.uk/bitesize/ks3/science/chemical_material_behaviour/compounds_mixtures/revision/1/
Task 16
Try this Quiz online to ensure your understanding
http://quizlet.com/11278379/jc-science-chemistry-element-compound-mixture-check-list-chap-19-flash-cards/
Task 17
Go to this website and complete the worksheet
http://chemsite.lsrhs.net/Intro/handouts/Elements_compounds_mixtures.pdf
Interpret chemical formulae:
A chemical formulae tells you what elements are in the chemical, also the ratio of each element in the chemical.
For example the formula for carbon monoxide is CO. It tells you that each molecule of carbon monoxide consists of one carbon atom joined to one oxygen atom.
Take care when writing your symbols and formulae. Be careful about when to use capital letters. For example CO means a molecule of carbon
monoxide but Co is the symbol for cobalt.
Formula and formulae
The word 'formulae' ("form-u-lee") is the plural of 'formula'. If we have more than one formula, we don't say formulas, we say formulae.
Numbers in formulae
If the molecule contains more than one atom of an element we use numbers to show this. The numbers are written to the right and slightly below
the element symbol. For example, the formula for carbon dioxide is CO2 . It tells you that each molecule has one carbon atom and two oxygen atoms.
Take care when writing these formulae. The small number is written at the bottom right corner.
Some formulae are more complicated. For example, the formula for sodium sulphate is Na2SO4 .
It tells you that sodium sulphate contains two sodium atoms (Na2 ), one sulphur atom (S) and four oxygen atoms (O4 ).
Task 18
Try these exercises:
A molecule of sulphur dioxide (SO2) contains……….one sulphur atom and two oxygen atoms
A compound of sodium sulphide (Na2S) contains……… X
A compound of iron oxide (Fe2O3) contains……… two iron atoms and three oxygen atoms
A compound of potassium hydroxide (KOH) contains………. X
A molecule of nitric acid (HNO3) contains…….. X
A compound of silver nitrate (AgNO3) contains……… X
A molecule of sulphuric acid (H2SO4) contains……… X
A compound of barium hydroxide (Ba(OH)2) contains……… X
A molecule of glucose (C6H12O6) contains………. X
A molecule of ethanol (C2H5OH) contains………. X
Why do you think the chemical formula for ethanol is C2H5OH and not C2H6O? X
Sometimes we see brackets followed by a subscript in a formula, this means you times everything inside the bracket by the subscript.
For example, the 2 in (NH4)2CO3 means there are 2× 1 = 2 N atoms and 2× 4 = 8 H atoms.
Task 19
Complete the rest of this table:
Compound Formula Elements present
Hydrogen peroxide H2O2 2 H atoms and 2 O atoms
Aluminium hydroxide Al(OH)3 X
Copper sulfate CuSO4 X
X FeS X
Lead nitrate Pb(NO3)2 X
Ammonium carbonate (NH4)2CO3 X
Need help? You can Read through this website below.
http://www.bbc.co.uk/schools/gcsebitesize/science/ocr_gateway/chemical_concepts/fundamentalrev3.shtml
Distinguish between chemical and physical change
http://www.bbc.co.uk/bitesize/standard/chemistry/elementsandreactions/chemical_reactions/revision/1/
PHYSICAL REACTION
What is a physical reaction: X
CHEMICAL REACTION
What is a chemical reaction: X
Task 20
Give 3 examples of physical reactions:
X
X
X
Give 3 examples of chemical reactions:
X
X
X
Link observations to chemical or physical changes
http://www.bbc.co.uk/bitesize/standard/chemistry/elementsandreactions/chemical_reactions/revision/1/
Task 21
What are the signs that show a chemical change has occurred, list 6 of them (be careful as some signs can occur in physical changes too).
X
Which sign also occur in both chemical and physical changes:
X
List evidence for a chemical reaction occurring
Task 22
Try the quiz below
http://quizlet.com/26629337/physical-changes-chemical-changes-pracitice-flash-cards/
Task 23
Is this a chemical or physical change?
A compound of sodium sulphide (Na2S) contains……… X
A compound of iron oxide (Fe2O3) contains……… two iron atoms and three oxygen atoms
A compound of potassium hydroxide (KOH) contains………. X
A molecule of nitric acid (HNO3) contains…….. X
A compound of silver nitrate (AgNO3) contains……… X
A molecule of sulphuric acid (H2SO4) contains……… X
A compound of barium hydroxide (Ba(OH)2) contains……… X
A molecule of glucose (C6H12O6) contains………. X
A molecule of ethanol (C2H5OH) contains………. X
Why do you think the chemical formula for ethanol is C2H5OH and not C2H6O? X
Sometimes we see brackets followed by a subscript in a formula, this means you times everything inside the bracket by the subscript.
For example, the 2 in (NH4)2CO3 means there are 2× 1 = 2 N atoms and 2× 4 = 8 H atoms.
Task 19
Complete the rest of this table:
Compound Formula Elements present
Hydrogen peroxide H2O2 2 H atoms and 2 O atoms
Aluminium hydroxide Al(OH)3 X
Copper sulfate CuSO4 X
X FeS X
Lead nitrate Pb(NO3)2 X
Ammonium carbonate (NH4)2CO3 X
Need help? You can Read through this website below.
http://www.bbc.co.uk/schools/gcsebitesize/science/ocr_gateway/chemical_concepts/fundamentalrev3.shtml
Distinguish between chemical and physical change
http://www.bbc.co.uk/bitesize/standard/chemistry/elementsandreactions/chemical_reactions/revision/1/
PHYSICAL REACTION
What is a physical reaction: X
CHEMICAL REACTION
What is a chemical reaction: X
Task 20
Give 3 examples of physical reactions:
X
X
X
Give 3 examples of chemical reactions:
X
X
X
Link observations to chemical or physical changes
http://www.bbc.co.uk/bitesize/standard/chemistry/elementsandreactions/chemical_reactions/revision/1/
Task 21
What are the signs that show a chemical change has occurred, list 6 of them (be careful as some signs can occur in physical changes too).
X
Which sign also occur in both chemical and physical changes:
X
List evidence for a chemical reaction occurring
Task 22
Try the quiz below
http://quizlet.com/26629337/physical-changes-chemical-changes-pracitice-flash-cards/
Task 23
Is this a chemical or physical change?
Explain X
Task 24
Test your knowledge so far: go to the following site to try out some quizzes
http://www.softschools.com/matching_games/science/5th_grade_matter/1076/
Task 25
Try this online Quiz
http://www.edinformatics.com/math_science/a_p_chem.htm
Identify the reactants and products for a chemical reaction
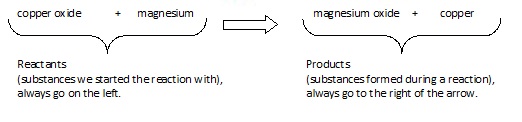
Reactants and Products
The chemicals we have at the start of a reaction are called reactants. When a chemical reaction takes place the atoms in these substances are rearranged to form new chemicals. The substances formed in a reaction are called products.
Example
If copper oxide is heated with magnesium it will react to form magnesium oxide and copper. We can write this as a simple word equation:
Task 24
Test your knowledge so far: go to the following site to try out some quizzes
http://www.softschools.com/matching_games/science/5th_grade_matter/1076/
Task 25
Try this online Quiz
http://www.edinformatics.com/math_science/a_p_chem.htm
Identify the reactants and products for a chemical reaction
Reactants and Products
The chemicals we have at the start of a reaction are called reactants. When a chemical reaction takes place the atoms in these substances are rearranged to form new chemicals. The substances formed in a reaction are called products.
Example
If copper oxide is heated with magnesium it will react to form magnesium oxide and copper. We can write this as a simple word equation:
Write simple word and formula equations for common reactions
Task 26
For each of the chemical reactions below:
Highlight or circle the reactants in one colour and the products in another colour, (colour in the boxes in the key, so you can remember which is which!)
Reactants =colour Products = colour
Task 27
Complete the word equation for each reaction, reaction one is done for you:
Reaction One
Sam mixed together some eggs, milk and flour. When he heated them in a large pan they reacted to form a big pancake!
eggs + milk + flour --> pancake
Reaction Two
When Katerina dropped a lump of magnesium into a test tube of hydrochloric acid, the chemical magnesium chloride was formed. Katerina also noticed a gas being formed. When a lit splint was placed in the gas it made a squeaky ‘pop’ sound.
X + Magnesium --> Magnesium chloride + X
Reaction Three
Luke reacted a lump of calcium carbonate with some sulphuric acid. Two of the products formed were water and calcium sulphate. The other product formed was a gas that turned lime water milky.
X + Calcium carbonate --> X + X dioxide
Reaction Four
Bradley heated some copper turnings with some sulphur powder very strongly in a test tube. He noticed that at the end of the experiment, there was a bluish colour left on the surface of the copper.
X + Sulphur --> X sulphide
Reaction Five
Chelsea was given some nitric acid and some iron oxide. She reacts these together and finds that she makes a solution of iron nitrate and iron water.
X + iron X --> X + X
Reaction Six
Milos put a brand new iron nail outside and studied it for several weeks. He noticed that gradually the surface of the nail
became coated with rust. He read in a science book that the chemical name for rust is “iron oxide”.
X + X --> X
Reaction Seven
Shona was learning about photosynthesis. She looked on the internet and found that in this process, plants take in carbon dioxide gas from the air and water from the ground. Using energy from the sun they can turn these chemicals into a kind of sugar called glucose and the gas oxygen.
X + X --> X + X
Reaction Eight
Jourdan told Shona that plants and animals respire. In this reaction they use up glucose and oxygen and make carbon dioxide and water and release energy that they can use to stay alive.
X + X --> X + X + ENERGY
Recall the three gas tests
Task 28
Three important gases:
Fill in a summary for these important gases
Hydrogen (H2)
properties X
uses X
Equation of making the gas
Oxygen (O2)
properties X
uses X
Equation of making the gas
Carbon dioxide (CO2)
properties X
uses X
Equation of making the gas
Gas tests
Task 29
Watch this video http://www.youtube.com/watch?v=LiAvDpl5aJA
Summarise what you saw
X
H2
Test ( with a picture) X
Observation for positive test X
O2
Test ( with a picture) X
Observation for positive test X
CO2
Test ( with a picture) X
Observation for positive test X
If you have finished and would like some extension work, try these:
http://quizlet.com/8927231/non-metals-flash-cards/
http://www.softschools.com/quizzes/science/physical_chemical_changes/quiz382.html
http://chemistry.about.com/od/testsquizzes/l/blatomsperiodicquiz.htm
http://www.sciencegeek.net/Chemistry/taters/Unit1AtomicStructure.htm
http://www.tutorvista.com/content/science/science-i/structure-atom/multiple-choice.php
http://quizlet.com/14011471/chemical-and-physical-changes-flash-cards/
http://quizlet.com/14378861/atoms-flash-cards/
Task 26
For each of the chemical reactions below:
Highlight or circle the reactants in one colour and the products in another colour, (colour in the boxes in the key, so you can remember which is which!)
Reactants =colour Products = colour
Task 27
Complete the word equation for each reaction, reaction one is done for you:
Reaction One
Sam mixed together some eggs, milk and flour. When he heated them in a large pan they reacted to form a big pancake!
eggs + milk + flour --> pancake
Reaction Two
When Katerina dropped a lump of magnesium into a test tube of hydrochloric acid, the chemical magnesium chloride was formed. Katerina also noticed a gas being formed. When a lit splint was placed in the gas it made a squeaky ‘pop’ sound.
X + Magnesium --> Magnesium chloride + X
Reaction Three
Luke reacted a lump of calcium carbonate with some sulphuric acid. Two of the products formed were water and calcium sulphate. The other product formed was a gas that turned lime water milky.
X + Calcium carbonate --> X + X dioxide
Reaction Four
Bradley heated some copper turnings with some sulphur powder very strongly in a test tube. He noticed that at the end of the experiment, there was a bluish colour left on the surface of the copper.
X + Sulphur --> X sulphide
Reaction Five
Chelsea was given some nitric acid and some iron oxide. She reacts these together and finds that she makes a solution of iron nitrate and iron water.
X + iron X --> X + X
Reaction Six
Milos put a brand new iron nail outside and studied it for several weeks. He noticed that gradually the surface of the nail
became coated with rust. He read in a science book that the chemical name for rust is “iron oxide”.
X + X --> X
Reaction Seven
Shona was learning about photosynthesis. She looked on the internet and found that in this process, plants take in carbon dioxide gas from the air and water from the ground. Using energy from the sun they can turn these chemicals into a kind of sugar called glucose and the gas oxygen.
X + X --> X + X
Reaction Eight
Jourdan told Shona that plants and animals respire. In this reaction they use up glucose and oxygen and make carbon dioxide and water and release energy that they can use to stay alive.
X + X --> X + X + ENERGY
Recall the three gas tests
Task 28
Three important gases:
Fill in a summary for these important gases
Hydrogen (H2)
properties X
uses X
Equation of making the gas
Oxygen (O2)
properties X
uses X
Equation of making the gas
Carbon dioxide (CO2)
properties X
uses X
Equation of making the gas
Gas tests
Task 29
Watch this video http://www.youtube.com/watch?v=LiAvDpl5aJA
Summarise what you saw
X
H2
Test ( with a picture) X
Observation for positive test X
O2
Test ( with a picture) X
Observation for positive test X
CO2
Test ( with a picture) X
Observation for positive test X
If you have finished and would like some extension work, try these:
http://quizlet.com/8927231/non-metals-flash-cards/
http://www.softschools.com/quizzes/science/physical_chemical_changes/quiz382.html
http://chemistry.about.com/od/testsquizzes/l/blatomsperiodicquiz.htm
http://www.sciencegeek.net/Chemistry/taters/Unit1AtomicStructure.htm
http://www.tutorvista.com/content/science/science-i/structure-atom/multiple-choice.php
http://quizlet.com/14011471/chemical-and-physical-changes-flash-cards/
http://quizlet.com/14378861/atoms-flash-cards/