Particle Theory of Matter
Describe the kinetic theory of matter
Reference: SW9 Pg 46-49
Task 8
Go to http://library.thinkquest.org/C0110228/molecules/kinetic.htm
and http://everythingscience.co.za/grade-10/03-states-of-matter/03-states-of-matter-02.cnxmlplus
Summarise the main points about the kinetic theory of matter
X
Go to http://www.youtube.com/watch?v=Y8j6peP5nNg which is a youtube video (5 mins) to help explain the kinetic theory of matter
Go to http://phet.colorado.edu/en/simulation/states-of-matter-basics which is an excellent for simulations (as shown in the youtube video
above)
Task 9
Practice writing long answers:
put your ideas together about solid/ liquid/gas in to 3 paragraphs.. The solid part is done for you – you need to write descriptions for the liquid and gas
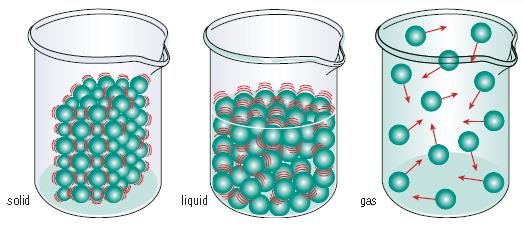
Solid
Particles are arranged in a regular way and are very close together. There are strong attractive forces holding the particles together in a fixed position. The particles have a small amount of energy and can only vibrate. They are unable to move freely.
Liquid
X
Gas
X
Temperature of the substance is related to the average X (two words) of particles.
For particles to theoretically stop moving, they have to be cooled to -273.15°C. This temperature is called X (two words)
For experts – go to http://www.youtube.com/watch?v=28F_oPDZHSk and http://www.youtube.com/watch?v=TNUDBdv3jWI (this will NOT be tested)
Link the kinetic theory of matter to the three states of matter
Change of states
When substances are heated or cooled, a change of state may occur.
This is a physical change as a new substance has not been formed, a physical property of the substance has changed.
We can use particle theory to explain changes of state.
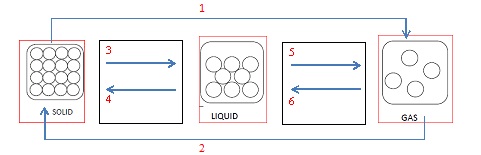
Task 10
Fill in the process names:
Reference: SW9 Pg 46-49
Task 8
Go to http://library.thinkquest.org/C0110228/molecules/kinetic.htm
and http://everythingscience.co.za/grade-10/03-states-of-matter/03-states-of-matter-02.cnxmlplus
Summarise the main points about the kinetic theory of matter
X
Go to http://www.youtube.com/watch?v=Y8j6peP5nNg which is a youtube video (5 mins) to help explain the kinetic theory of matter
Go to http://phet.colorado.edu/en/simulation/states-of-matter-basics which is an excellent for simulations (as shown in the youtube video
above)
Task 9
Practice writing long answers:
put your ideas together about solid/ liquid/gas in to 3 paragraphs.. The solid part is done for you – you need to write descriptions for the liquid and gas
Solid
Particles are arranged in a regular way and are very close together. There are strong attractive forces holding the particles together in a fixed position. The particles have a small amount of energy and can only vibrate. They are unable to move freely.
Liquid
X
Gas
X
Temperature of the substance is related to the average X (two words) of particles.
For particles to theoretically stop moving, they have to be cooled to -273.15°C. This temperature is called X (two words)
For experts – go to http://www.youtube.com/watch?v=28F_oPDZHSk and http://www.youtube.com/watch?v=TNUDBdv3jWI (this will NOT be tested)
Link the kinetic theory of matter to the three states of matter
Change of states
When substances are heated or cooled, a change of state may occur.
This is a physical change as a new substance has not been formed, a physical property of the substance has changed.
We can use particle theory to explain changes of state.
Task 10
Fill in the process names:
1:
2:
3:
4:
5:
6:
http://www.docbrown.info/page03/3_52states/3_52statesQ.htm
Explaining melting (solid-liquid) in terms of the kinetic theory of matter
When a solid is heated the particles gain more kinetic energy and vibrate faster.
At the melting point the particles have gained so much energy and vibrate so much that they are moving fast enough to overcome the forces of attraction between the particles and move far enough apart so that they can slide past each other.
When this happens the solid becomes a liquid.
Task 11
Explain what happens when water boils. (the quality of answer required is modelled above)
In your answer include the concepts of moving particles, energy, forces of attraction, distance between particles.
X
Task 12
Experiment on heating ice until it boils, see textbook SW9 page 49+50
Perform the experiment
Complete the table:
Time (min) 0 0.5 1.0 1.5 2.0 2.5 3.0 3.5 4.0 4.5 5.0
Temp(0C)
Graph your results
Answer the ‘Questions and Conclusions’ and the ‘Challenge’ on page 50
X
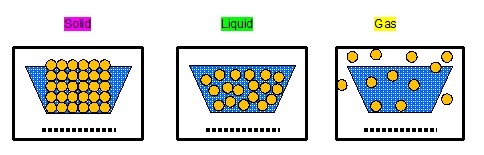
States of Matter Task: use 3 colours to group statements about the three states of matter.
Task 13
One statement in each column highlighted in magenta is done for you - there are other characteristics that also belong to the solid
state, highlight them in magenta as well.
Highlight all the statements about liquids in green
Highlight all the statements about gases in yellow
2:
3:
4:
5:
6:
http://www.docbrown.info/page03/3_52states/3_52statesQ.htm
Explaining melting (solid-liquid) in terms of the kinetic theory of matter
When a solid is heated the particles gain more kinetic energy and vibrate faster.
At the melting point the particles have gained so much energy and vibrate so much that they are moving fast enough to overcome the forces of attraction between the particles and move far enough apart so that they can slide past each other.
When this happens the solid becomes a liquid.
Task 11
Explain what happens when water boils. (the quality of answer required is modelled above)
In your answer include the concepts of moving particles, energy, forces of attraction, distance between particles.
X
Task 12
Experiment on heating ice until it boils, see textbook SW9 page 49+50
Perform the experiment
Complete the table:
Time (min) 0 0.5 1.0 1.5 2.0 2.5 3.0 3.5 4.0 4.5 5.0
Temp(0C)
Graph your results
Answer the ‘Questions and Conclusions’ and the ‘Challenge’ on page 50
X
States of Matter Task: use 3 colours to group statements about the three states of matter.
Task 13
One statement in each column highlighted in magenta is done for you - there are other characteristics that also belong to the solid
state, highlight them in magenta as well.
Highlight all the statements about liquids in green
Highlight all the statements about gases in yellow
Arranged in rows
Not able to change places
Spread to fill container
Takes on the shape of the container
Not arranged in any particular way
Fixed shape
Examples of Solids = ice, Table, Pencil,
Examples of Gases = Oxygen, Carbon dioxide, Sulfur dioxide,
Move about and change places
Relatively close together
Not held together as tightly as solids
Fixed volume
Can be easily compressed
Particles are far apart
Very weakly held together
Particles are held close together tightly
Particles Flow
Move very fast in all directions
Particles vibrate
Examples of Liquids = Water, Juice, Coffee
Not able to change places
Spread to fill container
Takes on the shape of the container
Not arranged in any particular way
Fixed shape
Examples of Solids = ice, Table, Pencil,
Examples of Gases = Oxygen, Carbon dioxide, Sulfur dioxide,
Move about and change places
Relatively close together
Not held together as tightly as solids
Fixed volume
Can be easily compressed
Particles are far apart
Very weakly held together
Particles are held close together tightly
Particles Flow
Move very fast in all directions
Particles vibrate
Examples of Liquids = Water, Juice, Coffee