Properties of Matter
Describe some physical properties of matter
Task 14
Watch the video /presentation, then complete the definitions
State
Colour
Density
Solubility
Malleability
Shape
Mass
Volume
Melting point
Boiling point
Conductivity
This is a song about physical properties: http://www.youtube.com/watch?v=uJOGy0dgmUU&list=PL1B5C9748DDFF1499
Task 15
Explaining the properties of matter in the three states of matter
Why do solids, liquids and gases have their own distinct properties? Let’s look at them at microscopic level.
Homework, please preview these websites before you come to class.
http://www.chem.purdue.edu/gchelp/liquids/character.html
http://www.youtube.com/watch?v=s-KvoVzukHo
http://www.bbc.co.uk/bitesize/ks3/science/chemical_material_behaviour/particle_model/revision/2/
Explaining properties:
Task 16
Put down your ideas about how particles in each state are arranged, the strength of attraction forces and how they move around.
The arrangement of the particles (regular or irregular)
Solid:
Liquid:
Gas:
The distance between particles (small or large)
Solid:
Liquid:
Gas:
Strength of attractive forces between particles (strong or weak)
Solid:
Liquid:
Gas:
Speed of particles (fast or slow)
Solid:
Liquid:
Gas:
Example in pictures
Solid
Liquid
Gas
Answer the following in small paragraphs:
1) Explain why air is able to be compressed? X
2) Explain why we can pour liquid and gas but not solid? X
Understand that density is mass per unit volume and use the formula density=mass/volume
Link density to floating and sinking.
Density is how much mass is packed in to a measured volume. Density tells us something about how tightly packed the particles are
in a substance. The following is a table showing the density of some substances.
Substance Density (g/cm3)
air 0.0013
cork 0.2
ice 0.9
water 1.0
aluminium 2.7
iron 7.8
lead 11.3
mercury 13.5
gold 19.3
A substance will float in water if its density is less than the density of water; while a substance will sink in water if its density is greater than the density of water
Name two substances that will float in water. X
Name two substances that will sink in water. X
Name a substance that will sink in water but float in mercury. X
Look up the average density of the human body X
Explain why the man in the photograph above is able to float in the Dead Sea (in the Middle East)? X
Task 14
Watch the video /presentation, then complete the definitions
State
Colour
Density
Solubility
Malleability
Shape
Mass
Volume
Melting point
Boiling point
Conductivity
This is a song about physical properties: http://www.youtube.com/watch?v=uJOGy0dgmUU&list=PL1B5C9748DDFF1499
Task 15
Explaining the properties of matter in the three states of matter
Why do solids, liquids and gases have their own distinct properties? Let’s look at them at microscopic level.
Homework, please preview these websites before you come to class.
http://www.chem.purdue.edu/gchelp/liquids/character.html
http://www.youtube.com/watch?v=s-KvoVzukHo
http://www.bbc.co.uk/bitesize/ks3/science/chemical_material_behaviour/particle_model/revision/2/
Explaining properties:
Task 16
Put down your ideas about how particles in each state are arranged, the strength of attraction forces and how they move around.
The arrangement of the particles (regular or irregular)
Solid:
Liquid:
Gas:
The distance between particles (small or large)
Solid:
Liquid:
Gas:
Strength of attractive forces between particles (strong or weak)
Solid:
Liquid:
Gas:
Speed of particles (fast or slow)
Solid:
Liquid:
Gas:
Example in pictures
Solid
Liquid
Gas
Answer the following in small paragraphs:
1) Explain why air is able to be compressed? X
2) Explain why we can pour liquid and gas but not solid? X
Understand that density is mass per unit volume and use the formula density=mass/volume
Link density to floating and sinking.
Density is how much mass is packed in to a measured volume. Density tells us something about how tightly packed the particles are
in a substance. The following is a table showing the density of some substances.
Substance Density (g/cm3)
air 0.0013
cork 0.2
ice 0.9
water 1.0
aluminium 2.7
iron 7.8
lead 11.3
mercury 13.5
gold 19.3
A substance will float in water if its density is less than the density of water; while a substance will sink in water if its density is greater than the density of water
Name two substances that will float in water. X
Name two substances that will sink in water. X
Name a substance that will sink in water but float in mercury. X
Look up the average density of the human body X
Explain why the man in the photograph above is able to float in the Dead Sea (in the Middle East)? X
Task 17
Explain why we use aluminium rather than iron to make air plane parts? X
Go to the simulation at http://phet.colorado.edu/en/simulation/density and investigate what is happening.
Calculating density
If we know the mass and the volume of a substance, we can calculate its density by using the formula
density = mass/volume
Task 18
Experiment on measuring density
Reference: SW9 page 41 Note that 1mL = 1 cm3
Object Mass (g) Initial volume (mL) Final volume (mL) Volume of object (mL) Density (g/cm3)
pebble 10.5 10.0 12.6mL 2.6 mL 4.04
X
X
X
When you do calculations you MUST:
1. Show the formula you are using
2. Show the numbers you are using
3. Calculate the answer
4. Give units for the answer
Task 19
1) If you know that a small piece of gold has a mass of 20g and a density of 19.3g/cm3, calculate its volume.
X
2) If you know that a small piece of aluminium has a volume of 50cm3 and a density of 19.3 g/cm3, calculate its mass.
X
Explain expansion and contraction as a change in density
Link expansion and contraction of a substance to heating and cooling, the kinetic theory of matter and the three states of matter
Expansion & Contraction
Reference: SW9 Pg 55
Substances expand or get bigger when they are heated up.
Substances contract or get smaller when they are cooled down. This property can be useful e.g. the liquid in thermometers expands and occupies more space, so the liquid rises up the thermometer.
Materials expanding and contracting can also cause problems e.g. railway lines expand in the summer heat and need special joints to stop them bending out of shape.
Explain why we use aluminium rather than iron to make air plane parts? X
Go to the simulation at http://phet.colorado.edu/en/simulation/density and investigate what is happening.
Calculating density
If we know the mass and the volume of a substance, we can calculate its density by using the formula
density = mass/volume
Task 18
Experiment on measuring density
Reference: SW9 page 41 Note that 1mL = 1 cm3
Object Mass (g) Initial volume (mL) Final volume (mL) Volume of object (mL) Density (g/cm3)
pebble 10.5 10.0 12.6mL 2.6 mL 4.04
X
X
X
When you do calculations you MUST:
1. Show the formula you are using
2. Show the numbers you are using
3. Calculate the answer
4. Give units for the answer
Task 19
1) If you know that a small piece of gold has a mass of 20g and a density of 19.3g/cm3, calculate its volume.
X
2) If you know that a small piece of aluminium has a volume of 50cm3 and a density of 19.3 g/cm3, calculate its mass.
X
Explain expansion and contraction as a change in density
Link expansion and contraction of a substance to heating and cooling, the kinetic theory of matter and the three states of matter
Expansion & Contraction
Reference: SW9 Pg 55
Substances expand or get bigger when they are heated up.
Substances contract or get smaller when they are cooled down. This property can be useful e.g. the liquid in thermometers expands and occupies more space, so the liquid rises up the thermometer.
Materials expanding and contracting can also cause problems e.g. railway lines expand in the summer heat and need special joints to stop them bending out of shape.
What do the particles do
When substances expand or contract, their particles stay the same size. It is the space between the particles that changes:
The particles in a solid vibrate more when it is heated, and take up more room
The particles in a liquid move around each other more when it is heated, and take up more room.
The particles in a gas move more quickly in all directions when it is heated, and take up more room.
Solids, liquids & gases all expand (get larger) when heated. When they are cooled they contract (get smaller).
As a solid is heated the particles gain X and vibrate more violently and start to bump in to each other. This causes them to move further X so that they have more X, therefore the solid as a whole X.
When a solid expands, the mass stays the same, but the X increases, therefore what will happen to the density? X
When the solid is cooled the particles lose e and vibrate more slowly. This causes them to slow down, move X together and occupy less X, therefore the solid as a whole X.
When a solid contracts, the mass stays the same but the X decreases, therefore what will happen to the density? X
Water is an exception !! - find out about the expansion of water. X
Explain some uses and problems associated with expansion and contraction
Look at the following pictures pictures – the harbour bridge (a metal bridge), a join in the structure of the harbour bridge and power lines in
summer.
When substances expand or contract, their particles stay the same size. It is the space between the particles that changes:
The particles in a solid vibrate more when it is heated, and take up more room
The particles in a liquid move around each other more when it is heated, and take up more room.
The particles in a gas move more quickly in all directions when it is heated, and take up more room.
Solids, liquids & gases all expand (get larger) when heated. When they are cooled they contract (get smaller).
As a solid is heated the particles gain X and vibrate more violently and start to bump in to each other. This causes them to move further X so that they have more X, therefore the solid as a whole X.
When a solid expands, the mass stays the same, but the X increases, therefore what will happen to the density? X
When the solid is cooled the particles lose e and vibrate more slowly. This causes them to slow down, move X together and occupy less X, therefore the solid as a whole X.
When a solid contracts, the mass stays the same but the X decreases, therefore what will happen to the density? X
Water is an exception !! - find out about the expansion of water. X
Explain some uses and problems associated with expansion and contraction
Look at the following pictures pictures – the harbour bridge (a metal bridge), a join in the structure of the harbour bridge and power lines in
summer.
Task 20
1. Explain why the harbour bridge requires finger-like joins in the bridge structure. Explain what would happen in summer when it is hot and in winter when it is cold.
X
2. Explain why the power cables, which are made of metal, droop so much in summer when it is hot. Describe and explain what would happen in winter when it is cold.
X
Know that different substances expand/contract at different rates
Task 21
Experiment on Bimetallic strip (solid metals of different types):
Take a picture and insert here and then describe and explain your observation:
Experiment on ball and ring (solids):
Take a picture and insert here and then describe and explain your observation:
Demonstration of coloured water in a tube(liquid):
Take a picture and insert here and then describe and explain your observation:
Demonstration of balloon on top of flask (gas)
Take a picture and insert here and then describe and explain your observation:
Some interesting facts
1. When liquids and gases get hot they expand, just as solids do. When heated by the same amount, liquid expands about 10 times more than solids, and gases expand about 1000 times more than liquids.
2. Different substances expand and contract by different amount.
3. Water is an exception, when it is heated, it expands, but when it is frozen, it also expands.
4. All gases expand at the same rate
Task 22
Watch the collapsing can experiment - video http://www.youtube.com/watch?v=FjYB8hgkvOU
Explain how it works - Use particle theory, including particles/ kinetic energy/forces/density.
X
Watch videos about hot air balloons- video http://www.youtube.com/watch?v=Ev9xlnWseyw and http://www.youtube.com/watch?v=wBAes6MMa40
Explain how a hot air balloon can rise? Use particle theory, including particles/kinetic energy/forces/density. X
Link diffusion of a substance to the kinetic theory of matter
Reference: SW Pg 53, 54
Task 23
Definition: Diffusion is X
1. Explain why the harbour bridge requires finger-like joins in the bridge structure. Explain what would happen in summer when it is hot and in winter when it is cold.
X
2. Explain why the power cables, which are made of metal, droop so much in summer when it is hot. Describe and explain what would happen in winter when it is cold.
X
Know that different substances expand/contract at different rates
Task 21
Experiment on Bimetallic strip (solid metals of different types):
Take a picture and insert here and then describe and explain your observation:
Experiment on ball and ring (solids):
Take a picture and insert here and then describe and explain your observation:
Demonstration of coloured water in a tube(liquid):
Take a picture and insert here and then describe and explain your observation:
Demonstration of balloon on top of flask (gas)
Take a picture and insert here and then describe and explain your observation:
Some interesting facts
1. When liquids and gases get hot they expand, just as solids do. When heated by the same amount, liquid expands about 10 times more than solids, and gases expand about 1000 times more than liquids.
2. Different substances expand and contract by different amount.
3. Water is an exception, when it is heated, it expands, but when it is frozen, it also expands.
4. All gases expand at the same rate
Task 22
Watch the collapsing can experiment - video http://www.youtube.com/watch?v=FjYB8hgkvOU
Explain how it works - Use particle theory, including particles/ kinetic energy/forces/density.
X
Watch videos about hot air balloons- video http://www.youtube.com/watch?v=Ev9xlnWseyw and http://www.youtube.com/watch?v=wBAes6MMa40
Explain how a hot air balloon can rise? Use particle theory, including particles/kinetic energy/forces/density. X
Link diffusion of a substance to the kinetic theory of matter
Reference: SW Pg 53, 54
Task 23
Definition: Diffusion is X
Watch http://www.youtube.com/watch?v=VY0mZUDvbH4 and http://www.youtube.com/watch?v=KRLNDTmBFZY
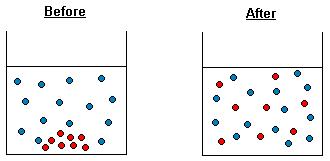
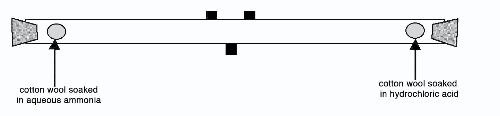
Demonstration of diffusion of gases (teacher only demo , hazardous substances used)
Demonstration of diffusion of gases (teacher only demo , hazardous substances used)
Watch http://www.youtube.com/watch?v=S9yuoLkfI3E and http://www.youtube.com/watch?v=L41KhBPBymA
Explain the formation of the ‘white smoke’
X
Which gas moved faster? X Explain your answer: X
Demonstration of diffusion in liquids (teacher only demo , leave for a few weeks)
Place some water in a large measuring cylinder. Add about 50mL of dilute acid and some universal indicator. Leave to settle. Then drop in large
crystals of sodium carbonate (washing soda) and drop in a little more acid carefully. Leave it to settle and observe every day
Take a photograph and place it here
Explain what is happening
X
Task 24
Experiment to show diffusion in liquids
Place some water in a large petri dish and let it become very still. Place a few crystals of potassium iodide at one side and a few crystals
of lead nitrate on the opposite side. Watch until something happens.
Take a photograph and place it here
Explain what has happened. X
In biology, we get diffusion of substances through membranes – the membranes have tiny holes in them through which the substance can move.
The movement of water through a semi-permeable membrane is given a special name - it is called X.
Watch http://www.youtube.com/watch?v=OXCKjhE1xco diffusion and osmosis in cells
Watch http://www.youtube.com/watch?v=wyJyDvQ6zaI to illustrate osmosis in raisins
Go to the simulation at http://phet.colorado.edu/en/simulation/membrane-channels and investigate what is happening.
Task 25
Experiments with Dry Ice (solid carbon dioxide)
Experiment 1:
Put dry ice in a balloon.
Observation and explanation: X
Experiment 2:
Put a piece of dry ice into a film canister.
Observation and explanation: X
Experiment 3:
Put a piece of dry ice into a beaker with a little water in it.
Observation and explanation: X
Experiment 4:
Pour the gas from experiment 3 into another beaker and put a lit splint into it.
Observation and explanation: X
Experiment 5:
Put some water into a beaker with a little bit of universal indicator in it along with a piece of dry ice.
Observation and explanation: X
Task 26
Practising comparison explanations:
Step 1: write a general statement (a topic sentence) about solids, liquids and gases.
X
Step 2: write a sentence or two comparing and contrasting the distance between particles.
X
Step 3: write a sentence or two comparing and contrasting the forces between particles.
X
Step 4: write a sentence or two comparing and contrasting the speed of the particles.
X
Task 27
Compare and contrast the difference between evaporating and boiling in a paragraph.
Use a contrasting linking word from here: (whereas, even though, but, while, less than, although, unlike, yet, however, different from, whilst…)
X
Explain the formation of the ‘white smoke’
X
Which gas moved faster? X Explain your answer: X
Demonstration of diffusion in liquids (teacher only demo , leave for a few weeks)
Place some water in a large measuring cylinder. Add about 50mL of dilute acid and some universal indicator. Leave to settle. Then drop in large
crystals of sodium carbonate (washing soda) and drop in a little more acid carefully. Leave it to settle and observe every day
Take a photograph and place it here
Explain what is happening
X
Task 24
Experiment to show diffusion in liquids
Place some water in a large petri dish and let it become very still. Place a few crystals of potassium iodide at one side and a few crystals
of lead nitrate on the opposite side. Watch until something happens.
Take a photograph and place it here
Explain what has happened. X
In biology, we get diffusion of substances through membranes – the membranes have tiny holes in them through which the substance can move.
The movement of water through a semi-permeable membrane is given a special name - it is called X.
Watch http://www.youtube.com/watch?v=OXCKjhE1xco diffusion and osmosis in cells
Watch http://www.youtube.com/watch?v=wyJyDvQ6zaI to illustrate osmosis in raisins
Go to the simulation at http://phet.colorado.edu/en/simulation/membrane-channels and investigate what is happening.
Task 25
Experiments with Dry Ice (solid carbon dioxide)
Experiment 1:
Put dry ice in a balloon.
Observation and explanation: X
Experiment 2:
Put a piece of dry ice into a film canister.
Observation and explanation: X
Experiment 3:
Put a piece of dry ice into a beaker with a little water in it.
Observation and explanation: X
Experiment 4:
Pour the gas from experiment 3 into another beaker and put a lit splint into it.
Observation and explanation: X
Experiment 5:
Put some water into a beaker with a little bit of universal indicator in it along with a piece of dry ice.
Observation and explanation: X
Task 26
Practising comparison explanations:
Step 1: write a general statement (a topic sentence) about solids, liquids and gases.
X
Step 2: write a sentence or two comparing and contrasting the distance between particles.
X
Step 3: write a sentence or two comparing and contrasting the forces between particles.
X
Step 4: write a sentence or two comparing and contrasting the speed of the particles.
X
Task 27
Compare and contrast the difference between evaporating and boiling in a paragraph.
Use a contrasting linking word from here: (whereas, even though, but, while, less than, although, unlike, yet, however, different from, whilst…)
X